|
Approximately one in every 25,000 individuals is diagnosed with NF2, and all NF2 patients will eventually experience profound hearing loss secondary to the tumor growth, radiation effects, and/or surgical resection of bilateral vestibular schwannomas (VS). The ABI was approved by the United States Food and Drug Administration (FDA) in 2000 for patients with Neurofibromatosis type 2 (NF2) and there are no audiologic criteria. Over one thousand ABIs have been placed in deaf adults and children around the world and is the most commonly placed surface stimulator in the central nervous system. The basic design of the ABI multichannel array, however, has not changed in several decades. Advancements have been made in the external speech processor and receiver stimulator technology but are otherwise identical to systems used for CIs. Unlike thin and flexible CI electrodes that follow the curved spatial arrangement of frequency selectivity in the cochlea (tonotopic axis), ABI electrodes are embedded in flat and rigid Silastic paddle that lies along the highly curved surface of the CN in the brainstem ( Guex et al., 2015). The external system consists of a battery source, microphone, speech processor, transmitter coil, and magnet worn above and behind the ear ( Figure 1A) the internal system consists of a surgically placed receiver-stimulator and magnet (in patients who require frequent magnetic resonance imaging (MRI), the magnet is removed and replaced with a metallic non-magnetic spacer at the time of ABI placement), a ground electrode, and an electrode array ( Figures 1B,C). Similar to a CI, an ABI consists of an external ear-level worn device and an internal receiver-stimulator implant. The first ABI was developed at the House Ear Institute (HEI) in the 1970s ( Edgerton et al., 1982). The ABI bypasses the cochlear nerve to electrically stimulate second order neurons in the cochlear nucleus (CN) using a multichannel surface array in patients with cochlear and retrocochlear pathologies. The auditory brainstem implant (ABI) is a neuroprosthetic device that provides hearing sensations to deaf patients who are ineligible for the cochlear implant (CI) due to anatomic constraints. 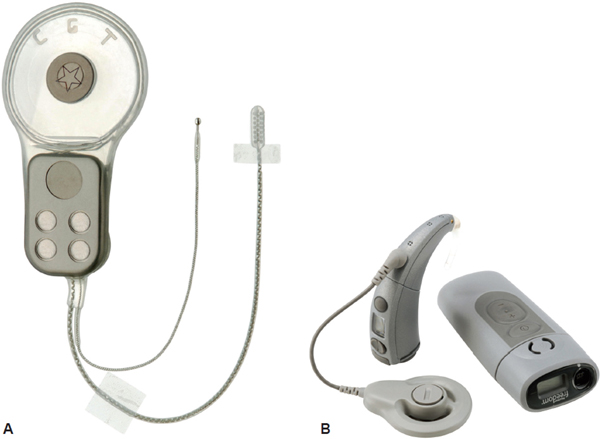
The aims of the following review are to describe the history of the ABI and innovations leading up to the modern ABI system, as well as highlight areas of future development in implant design. The auditory brainstem implant (ABI) was first developed nearly 40 years ago and provides auditory rehabilitation to patients who are deaf and ineligible for cochlear implant surgery due to abnormalities of the cochlea and cochlear nerve. 
5Department of Neurological Surgery, University Hospitals Cleveland Medical Center, Case Western Reserve University School of Medicine, Cleveland, OH, United States.4Center for Neuroprosthetics, École Polytechnique Fédérale de Lausanne, Geneva, Switzerland.3Department of Otology and Laryngology, Harvard Medical School, Boston, MA, United States.2Department of Otolaryngology, Massachusetts Eye and Ear, Boston, MA, United States.1Department of Otolaryngology, Icahn School of Medicine at Mount Sinai, New York, NY, United States. 
Kanumuri 2,3 Nicolas Vachicouras 4 Jonathan Miller 5 Stéphanie Lacour 4 M.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
